We will be in essence extending this concept here but now, the electrons will be moving in such a way as to create or destroy bonds. The resonance structures involved electrons jumping around at thousands of times the rate that the atoms themselves could move, so it was just simpler to assume the atoms don’t move. We have explored this concept earlier when we looked at resonance structures of a single molecule or polyatomic ion species. Remember that the electrons are thousands of times lighter than the nuclei of the atoms themselves so although we often think and speak as if it’s the atoms that do the moving, we are better off to imagine the electrons as doing the moving while the atoms are relatively motionless. To keep track of these changes, we use curly arrows to represent the movement of electrons. Many reactions take several steps to occur and each step may involve the creation or destruction of bonds. 
Generally, we can write an overall process as a simple balanced chemical reaction, but that reaction often does not happen in a single step as is implied by the single equation. When we draw out the reaction mechanisms, it’s important to keep track of where the electrons are, especially the ones involved in forming and breaking bonds. For example, a carbon-carbon double bond has two pairs of electrons in close proximity to each other, making for a concentration of negative charge whereas, if there is an electronegative atom bonded to a carbon, it will tend to inductively withdraw some of the electron density away from the carbon creating a positive charge concentration on that carbon. The charges in the substrate (the main organic molecule being reacted) will occur depending on the structure composition of the molecule.  
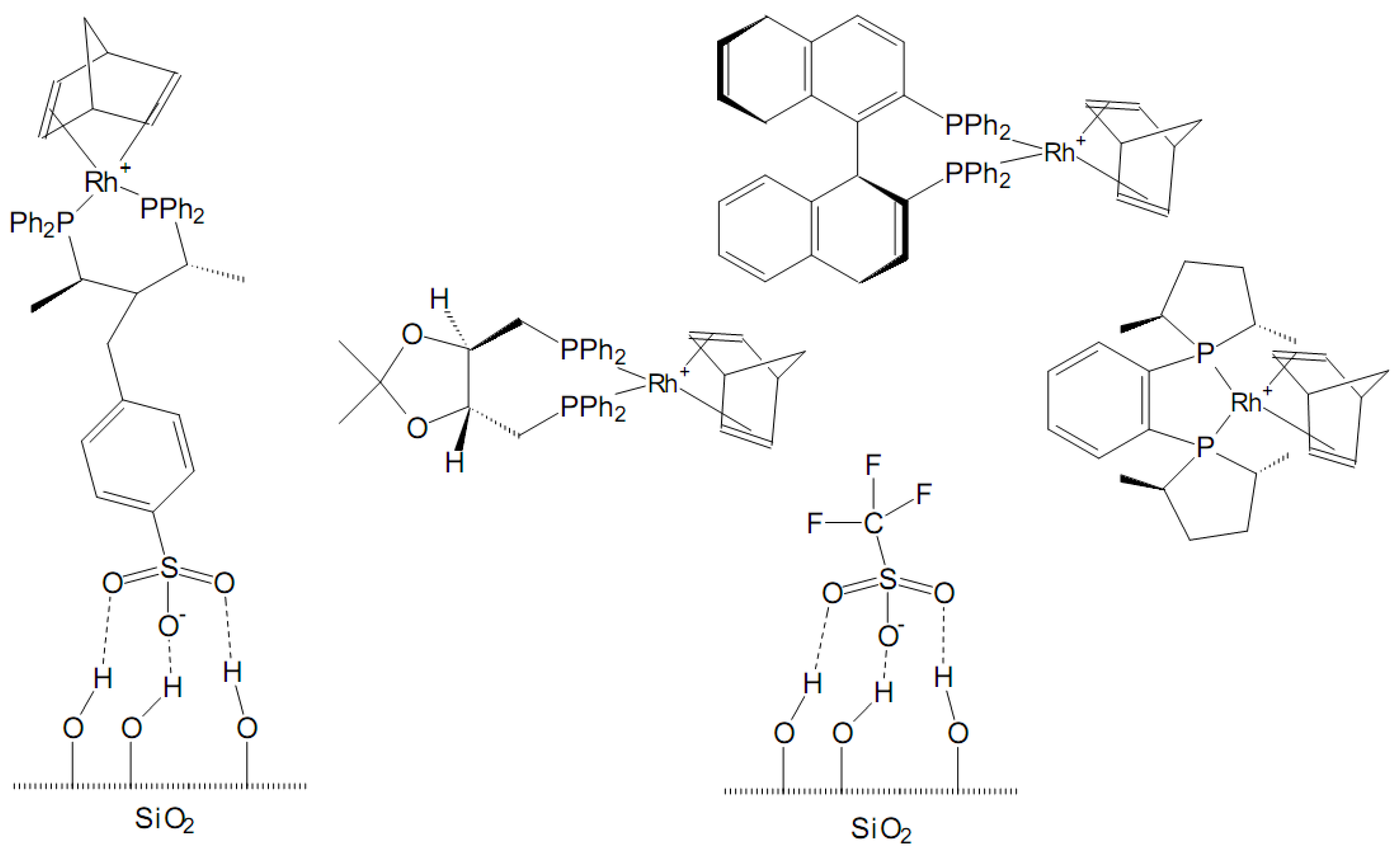
We also show that the rate of the Wittig reaction of ylide 2 with aldehyde 14 is significantly faster than the rate of either C- or O-attack calculated using lg k (20 ☌) = sN(E + N), thus indicating that the oxaphosphetane is formed by a concerted cycloaddition.Chemical reactions in organic compounds tend to occur at reactive sites on the molecules, where there is buildup of either positive or negative charge. It is shown that the principle of hard and soft acids and bases (HSAB) and the related Klopman-Salem concept of charge and orbital control lead to incorrect predictions of regioselectivity. The results derived from our investigations are employed to rationalize the behavior of ambident nucleophiles 1 and 2 in reactions with carbon-centered electrophiles in general. In other cases, rearrangement to the thermodynamically more stable products arising from C-attack occurs. However, the initially formed benzhydryloxyvinylphosphonium ions can only be observed by NMR spectroscopy when benzhydryl cations with high Lewis acidity are employed. Generally, O-attack of benzhydrylium ions is faster than C-attack. For some reactions, it was possible to determine the second-order rate constant for O-attack as well as for C-attack and to derive the nucleophile-specific parameters N and sN according to the correlation lg k (20 ☌) = sN(E + N) for both nucleophilic sites. As ambident nucleophiles, ylides 1 and 2 can react at oxygen as well as at the α-carbon. The kinetics and mechanism of the reactions of formyl-stabilized ylide Ph3P═CHCHO (1) and acetyl-stabilized ylide Ph3P═CHCOMe (2) with benzhydrylium ions (Ar2CH(+), 3) were investigated by UV-vis and NMR spectroscopy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
